
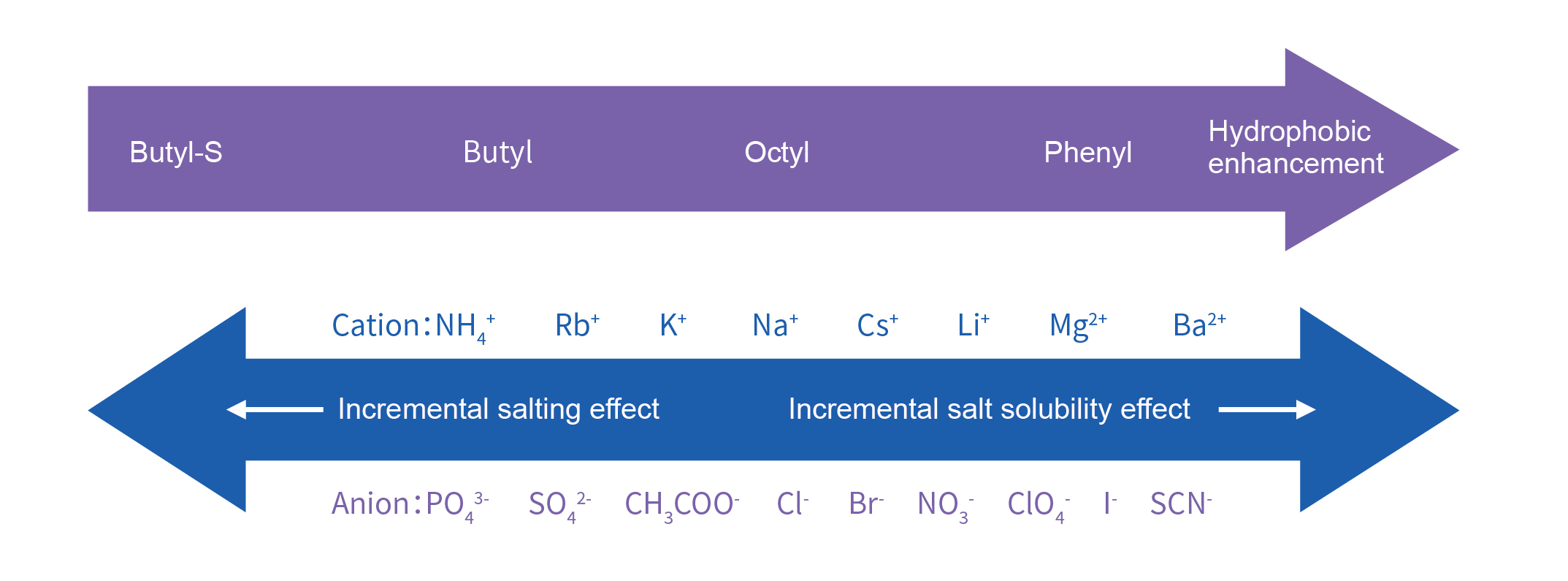
Hydrophobic interaction resins separates proteins based on differences in hydrophobicity, i.e., based on reversible interactions between proteins and hydrophobic groups on the surface of hydrophobic interaction resins. Hydrophobicity isenhanced at high ionic strengths and therefore binding in a high ionic strength environment is usually eluted by reducing the ionic strength. The unique adsorption separation mode makes hydrophobic interaction resins an ideal purification method after ammonium sulfate chromatography or after ion exchange high salt elution.
Tips:
★ The hydrophobic interaction strength varies with different ligands and ligand concentrations in the media.
★ Adjust the salt concentration in the buffer when performing hydrophobic chromatography for different proteins or using different media.
★ Temperature and pH significantly affect protein hydrophobicity. Maintain consistent pH and temperature during hydrophobic chromatography.
